Scientists have published a detailed ‘atlas’ of the placenta and uterus, showing how these unique tissues grow and evolve during pregnancy to match the developing fetus.
In creating this new map, scientists uncovered a previously undescribed cell subtype that appears to be unique to pregnancy.
you may like
“It was an exciting moment during the research,” study lead author Jingjing Li, an associate professor of neurology at UCSF who studies human genomics, said of the cell discovery. “We asked around and no one knew what they were.”
These newly described cells appear to be involved in linking the placenta to the maternal blood supply and have receptors that respond to cannabinoids. Cannabinoids include the cannabis compounds THC and CBD, as well as chemicals produced within the body. Researchers therefore think these cells may help explain why cannabis use during pregnancy is linked to health effects such as reduced blood flow to the placenta. Insufficient oxygen supply to the fetus. Increased risk of premature birth, low birth weight, and NICU admission.
The sensitivity of these cells to cannabinoids is unlikely to fully explain the risks posed by cannabis use during pregnancy, Lee told Live Science. Other potential culprits are described in the medical literature. Nevertheless, these newly discovered cells warrant further study, he said.
Significant “speed bump”
Prior to this new study, published April 8 in Nature, other research groups had used similar techniques to map the placenta and uterus. However, these previous studies covered only some chapters of pregnancy.
“The biggest difference is that we’re looking at the entire time course from early pregnancy to birth,” Lee said. The new atlas incorporates data from tissue collected from week 5 to week 39 of pregnancy and subsequently stored in tissue banks at UCSF and Stanford University.
In Lee’s lab, placental development is one of the team’s main research focuses, and the tissue is analyzed in detail at single-cell resolution. Their new atlas incorporates a snapshot of which genes were active and which proteins were present in the cells analyzed at specific stages of pregnancy. We will also look at “chromatin accessibility,” which reflects how DNA molecules are packaged within cells and which genes are activated at a given moment.
In total, the research team analyzed approximately 1.2 million placental and uterine cells. This included 200,000 isolated cells and 1 million cells implanted in their original location within the tissue.
What to read next
This study reveals an interesting link between the gene activity of specific cells and their behavior.
For example, during early pregnancy, certain fetal cells invade the uterus and its major arteries to help establish blood flow to the placenta. The researchers used machine learning to predict how deeply certain cells would invade the uterus based on gene activity. If this penetration fails, for example if the cells do not penetrate deep enough or penetrate too deeply, it can lead to complications such as pre-eclampsia and placenta accreta.
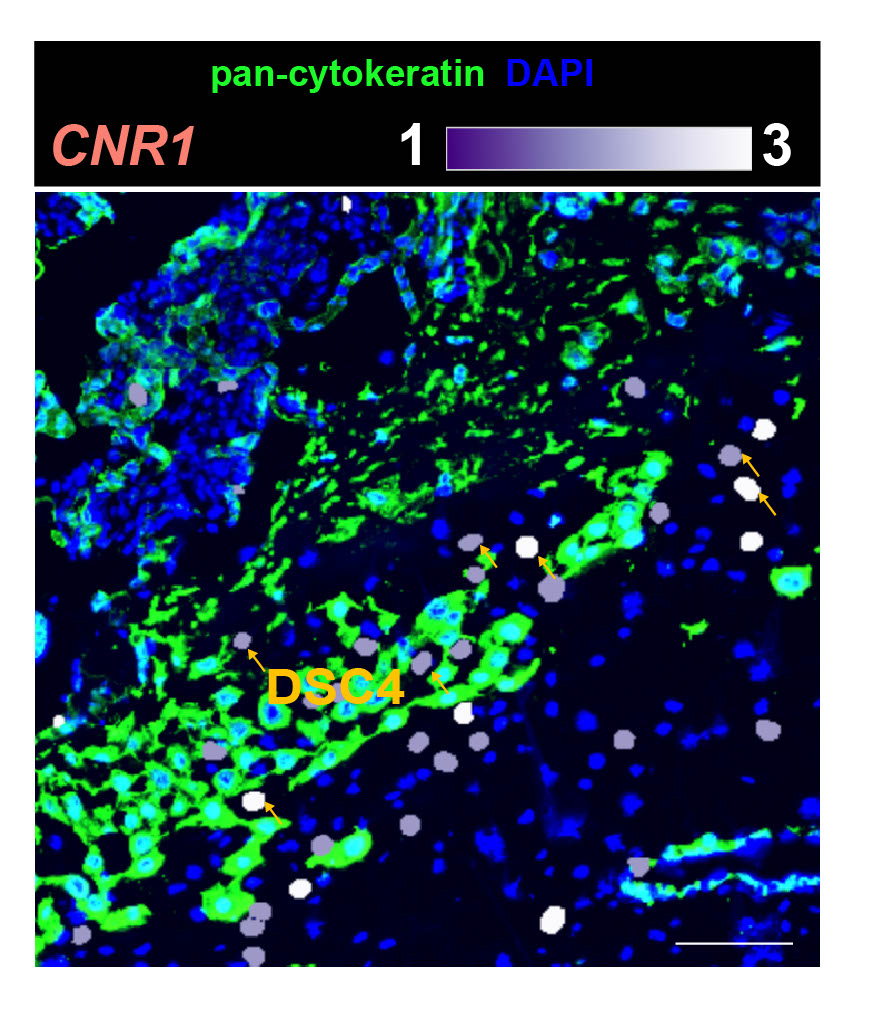
A new cell type identified by the researchers turns out to help regulate invasion. By sending specific signals, cell types act as “speed bumps” that prevent processes from proceeding too quickly, Lee said.
“It’s at the forefront of the maternal-fetal interface,” Wen told Live Science. He said the variety of proteins carried by these cells supports the idea that they regulate the behavior of other cells at this critical interface.
With the completed map in hand, the researchers combined their results with data from a vast body of genetic research on preeclampsia, premature birth, and miscarriage. These published studies have revealed associations between specific genetic variants and the risk of these complications. The team was then able to actively use these genes and therefore identify specific cells within the placenta and uterus that were most vulnerable to this condition.
“The question is, ‘In which cell types are these high-risk variants effective?'” Lee said. “This can help us learn which cells underlie those complications” and potentially develop treatments to target those cells in the future.
Although the study collected a large amount of data, Lee emphasized that there is still more work to be done. Because this study focused on healthy pregnancies, questions remain about how pregnancies affected by different conditions differ from this baseline. The team is now working with clinical partners to begin comparing them. Overall, the researchers aim to increase the total number of cells analyzed so that they can fully capture the cellular diversity within the pregnant uterus.
“Including more cells, more samples could lead to many new and exciting discoveries,” Lee said. “So this is really the starting point.”
This article is for informational purposes only and does not provide medical advice.
Source link