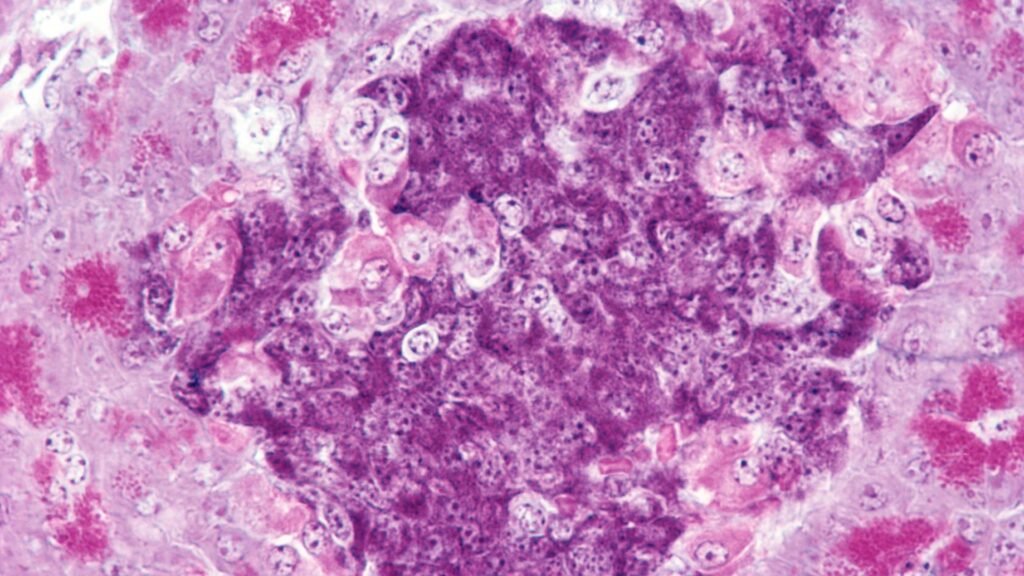
Scientists cured type 1 diabetes in mice without causing long-term immunosuppression.
Type 1 diabetes has historically required lifelong doses of powerful immunosuppressive drugs for the immune system to attack insulin-producing cells and replace them with transplanted cells from a donor, severely limiting the scope of such transplants.
you may like
More research is needed before this type of treatment is available to patients in the clinic, and mixed immune systems are difficult to balance. But if large follow-up trials in humans show that the transplant process is safe and durable, it could pave the way to reversing the potentially deadly disease.
“This is a way to potentially treat diabetes,” Dr. John DiPersio, an oncologist at Washington University in St. Louis who studies cell therapy but was not involved in the study, told Live Science. “In theory, this represents a huge step forward.”
Inducing intolerance
In type 1 diabetes, the immune system mistakenly attacks insulin-producing cells in the pancreas, or islets. People with this disease must take insulin for the rest of their lives because without insulin, blood sugar levels rise and eventually lead to death. Even with the best treatments, people with type 1 diabetes still suffer from high rates of complications such as heart disease, kidney disease, and eye damage.
Scientists have been trying to treat the disease for decades by replacing destroyed islands with new islands, such as those taken from corpses. However, to keep the body from attacking the transplanted cells, patients must take powerful immunosuppressive drugs for the rest of their lives. As a result, islet transplants are typically only performed in clinical trials or for patients who require another organ replacement, such as a kidney or liver transplant.
Using bone marrow stem cells and islet cells from the same donor may solve the problem of immune rejection. Stem cells transplanted into a special niche in the bone regenerate the white blood cells of the immune system. The newly regenerated immune system will have no cells to attack the islets and will recognize the transplanted islets as “self” rather than foreign.
When donors and recipients are mixed, the donor’s immune system, or blood system, can influence the recipient’s behavior. [immune cells] of the recipient.
Dr. Judith Sizzle, Professor, Stanford University School of Medicine
However, that process required removing the host’s own bone marrow stem cells. “It’s like a game of musical chairs,” study lead author Dr. Judith Sizzle, a professor at Stanford University School of Medicine, told Live Science. “If you don’t take the recipient stem cells out of the niche, you can’t take in the donor cells either.”
In the past, this process required chemotherapy and radiation treatments to completely eliminate the host’s immune system, leaving people vulnerable to infection for weeks.
What to read next
Shizuru’s team wondered if there was a less toxic therapy that could re-educate the host’s immune system rather than erase it. “When donors and recipients are mixed, the donor’s immune system, or blood system, can influence the patient’s behavior. [immune cells] It belongs to the recipient,” Shizuru said.
They devised a multistep process using multiple antibodies, low doses of radiation, and a rheumatoid arthritis drug called baricitinib, and tested the protocol on more than a dozen mice. This “conditioning” process of the immune system created space in the recipient’s bone marrow for some donor stem cells without erasing all of the recipient’s stem cells. They also suppressed various parts of the immune system long enough for the donor stem cells and islets to take hold.
This allowed the research team to transplant bone marrow stem cells and pancreatic islets from the same donor into recipient mice. As the donor stem cells matured, the cells educated the recipient’s remaining immune system to tolerate the foreign tissue. The mature mixed immune system also culled recipient cells trained to specifically attack pancreatic islets, thereby eliminating cells that promote autoimmunity. “The graft sticks and stays in place,” Shizuru said. “It’s here for the long term.”
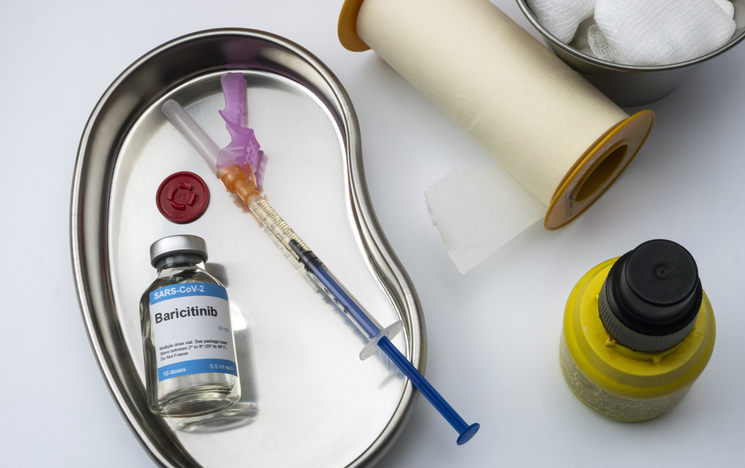
From start to finish, the process took about 12 days, the immune system was never completely wiped out, and the radiation doses were lower than those typically used in bone marrow transplants. “We created this [a] It’s a much gentler prescription,” Shizuru said.
After 20 weeks, the mice were still secreting insulin, and blood tests and post-mortem analysis showed that the mice’s immune systems were functioning well and did not reject the transplant, the study authors noted in a paper published in the January issue of the Journal of Clinical Investigation.
Still, many hurdles remain before this becomes a viable treatment in humans, said DiPersio, author of an accompanying commentary article in the same journal. First, some of the antibodies that worked in mice do not have approved analogs in humans, so this needs to be improved. Second, this method requires obtaining both bone marrow and islets from the same donor, the latter of which is already in short supply.
But the trickier problem is that building a mixed host-receptor immune system is a delicate balancing act, DiPersio says.
The researchers maintained this balance in mice, which typically live only a year or two.
For this process to result in healing, humans will need various immune system components to remain in balance for decades. “It’s difficult to do that over a long period of time,” DiPersio said. An imbalance can lead to gradual death of pancreatic islets or dangerous tissue rejection, he said.
This article is for informational purposes only and is not intended as medical advice.
Bagchandani, P., Ramos, S.A., Rodriguez, B., Gu, X., Pathak, S., Zhou, Y., Moon, Y., Nowlin, N., Zhang, C.A., Poyser, J., Velasco, B.J., Chao, W., Kwon, H., Rodriguez, R., Burgos, D.M., Miranda, M.A., Meyer, E., Shizuru, J.A., & Kim, S.K. (2025). Cure of autoimmune diabetes in mice by pancreatic islet and hematopoietic cell transplantation after CD117 antibody-based pretreatment. Journal of Clinical Research, 136(1). https://doi.org/10.1172/jci190034
Source link