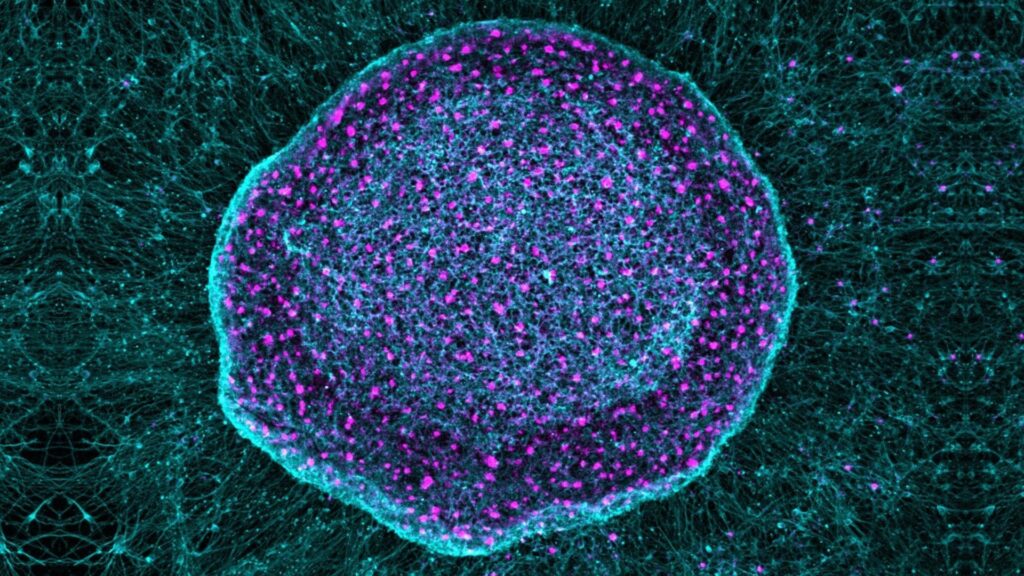
The experimental treatment uses specialized neurons derived from stem cells to “absorb” triggers of pain and inflammation in arthritic knees of mice.
The experiments in laboratory mice suggest that this treatment could help people with chronic pain caused by diseases such as osteoarthritis, for example. Researchers say a “pain sponge” could help patients stop relying on opioid drugs for pain relief.
you may like
“The potential for this treatment to reduce pain and slow cartilage degeneration is particularly compelling for osteoarthritis,” Chuan-Ju Liu, a professor of orthopedic surgery at Yale University who was not involved in the study, told Live Science.
How the pain sponge works
The treatment, known as SN101, uses human pluripotent stem cells (hPSCs), which can differentiate into any type of cell in the body. In the study, led by Gavsan Lee, professor of neurology at Johns Hopkins University School of Medicine, researchers engineered hPSCs to differentiate into specialized sensory neurons.
These neurons effectively acted as sponges for inflammatory pain signals. They isolated the signal before it was transmitted to the brain and caused pain.
In theory, the therapy could be effective for all types of chronic pain, said Daniel Saragnese, co-founder of SereNeuro Therapeutics, the biotechnology company developing SN101. However, researchers have so far only tested its effectiveness against osteoarthritis, the most common form of arthritis.
This degenerative disease is characterized by inflammation and chronic pain that affects the joints, primarily the hips, knees, hips, and neck. It causes pain and stiffness as well as inflammation caused by the destruction of bone, cartilage, and other tissues. There is no cure.
Currently, osteoarthritis symptoms are managed with physical therapy and lifestyle changes, including a variety of pain medications, including over-the-counter and topical pain relievers, opioids, and steroid injections.
In the context of neurodegenerative diseases such as multiple sclerosis, Alzheimer’s disease, and Parkinson’s disease, scientists have been working on using hPSCs to replace or repair damaged neurons. But with SN101, the researchers are taking a different approach. New hPSC-derived neurons are injected into the site of inflammation and coexist with other pain-sensing neurons rather than replacing them.
you may like
The new neurons act as biological decoys, binding to nearby inflammatory factors before being captured by the body’s original neurons.
Potential advantages of SN101
Chronic pain is defined as pain that lasts more than three months and is often managed with opioid drugs, which bind to receptors in the body and reduce the intensity of pain. However, opioids can cause unwanted side effects such as nausea and vomiting, and carry the risk of addiction.
Despite the downsides, it is estimated that approximately 9% of patients with knee osteoarthritis rely on opioids, which can lead to excessive long-term use. That’s why scientists are constantly searching for safer and more efficient pain management techniques.
By using biologically complex cells that naturally express multiple pain receptors, SN101 may more closely reflect how pain and inflammation manifest in living tissues, Liu said. This may help eliminate the cause of the pain. Opioids, on the other hand, bind to receptors in the brain and temporarily block painful sensations, preventing them from reaching the signals at the root of the pain.
“However, this research is still in the preclinical stage,” Liu emphasized.
The research must pass important milestones, including formal toxicity studies, long-term safety evaluations and first-in-human clinical trials, before it can be used in humans, he said. Nevertheless, he called the idea behind the therapy “revolutionary.”
In a recent study, researchers pointed to several limitations that need to be investigated before SN101 is deemed safe for humans. One is the immunogenicity of the treatment, that is, whether it causes an adverse immune response in the body. Another limitation is that the knee joints of humans and mice are very different, so some of the findings in arthritic mice may not apply to humans.
“Human joints are large. [than mouse joints]”It’s more mechanically complex and exposed to cumulative stress over decades,” Liu said. Additionally, he said, “Pain processing and immune-neuronal interactions can differ significantly between mice and humans, which may impact both therapeutic efficacy and durability.”
Ectopic engraftment of hPSC-derived nociceptive neurons for analgesia and joint homeostasis. Zhuolun Wang, Weixin Zhang, Ju Wang, Zhiping Wu, Xu Cao, Junmin Peng, Gabsang Lee, Xinzhong Dong. BioRxiv 2025.12.16.694733;doi: https://doi.org/10.64898/2025.12.16.694733
Source link