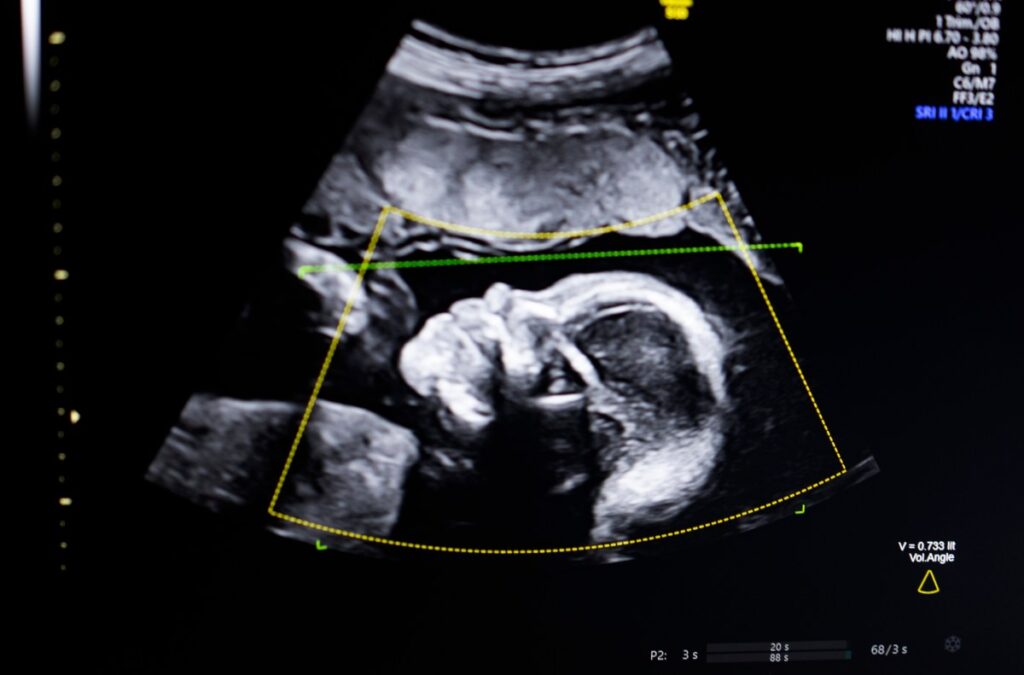
BioticsAI, winner of TechCrunch Disrupt Battlefield 2023, announced Monday that it has received FDA clearance for its AI software that helps detect fetal abnormalities from ultrasound images.
The product was conceived by founder and CEO Robbie Bustami, who grew up in a family of obstetricians, including his mother, aunt, and uncle. He spent a lot of time in hospitals during his childhood and often traveled with his mother as she cared for him across the United States.
After studying coding and studying computer science at the University of California, Irvine, Bustami launched BioticsAI in 2021 in collaboration with Salman Khan, Chaskin Salov, and Dr. Hisham El-Gamal.
The technology uses computer vision AI to “support fetal ultrasound quality assessment, anatomical integrity, automated reporting, and seamless integration into clinical workflows,” Bustami told TechCrunch.
He hopes his technology will help combat the fact that the United States has some of the worst prenatal outcomes for mothers among high-income countries. Black women in particular face extremely high maternal mortality rates;
Bustami said prenatal ultrasounds are the “cornerstone” of pregnancy monitoring, but poor image quality can lead to misdiagnoses.
Bustami said the biggest challenge was not building an AI model trained on a diverse set of hundreds of thousands of ultrasounds, but ensuring the technology would work reliably in the real world, particularly in the demographics most at risk of a tragic outcome.
tech crunch event
san francisco
|
October 13-15, 2026
“In an environment where disparities in health care outcomes are well documented, it was important to show consistent performance not just in the ideal case but across subgroups of patients,” continued Bastami.
The CEO said it took just under three years to go through the FDA process, which includes testing and validating the product. This experience taught him and his team how important it is to have engineering, product, clinical, and regulatory operations closely aligned from the beginning. “By designing the product, clinical validation, and regulatory pathway together rather than sequentially, we were able to move quickly,” he said.
Now that it has received FDA approval, Bustami said the company’s next focus is to expand to various health systems across the country. He also has plans to add more capabilities for fetal medicine and reproductive health.
“We are well-positioned to expand both distribution and clinical impact while continuing to deepen the power of our technology,” he said.
This article has been updated to correct the spelling of the co-founder’s name.
Source link